Lab case study: Love science, hate plastic waste

Andy Arnott, Projects Coordinator (Labs), summarises how biohazard waste was reduced by 40% in a pioneering study by a lab in the University of Edinburgh Roslin Institute.
The pilot study
Researchers in a microbiology lab in the University of Edinburgh’s Roslin Institute (Veterinary) have undertaken a pilot study to find out how they can reduce the amount of plastic waste produced by their lab. The Laboratory of Bacterial Evolution and Pathogenesis is small, with only seven researchers. Given the success of this pilot, it is exciting to consider the potential of these behaviours being adopted by larger labs more widely.
Life sciences laboratories typically use a lot of single-use plastic items to handle and manipulate biological materials. Once used and contaminated, these items need to be disposed of as biohazard waste – unless they can be decontaminated and reused.
Using the waste hierarchy, they have tried to reduce purchases and reuse items. Currently, recycling is not an option at the University of Edinburgh for used lab plastics. However, we are working with our waste contractor, and colleagues from other Higher Education Institutions across Scotland, to investigate if this can change in future.
Impressive savings of materials and money
The work is ongoing, but so far has delivered a reduction of 40% on the weight of materials sent for disposal as biological waste. The team shared promising stats for specific items:
- The transition to reusable metal inoculation loops saved 1,300 plastic inoculation loops over a 4-week period, the equivalent of 15,600 plastic loops per year.
- Over the first 4-week test period we saved approximately 1,670 plastic tubes, the equivalent of 20,000 plastic tubes per year. This is a saving of £1,390 per year.
- By reusing plastic cuvettes, which are purchased in boxes (each with 100 units), no new boxes of plastic cuvettes were purchased over a 4-month period, whereas five boxes of cuvettes were purchased in the 4-month period preceding the test period.
Overall, these savings in terms of the number of plastic items led to a reduction of laboratory waste of 43 kg in a 4-week period (54 vs 97 kg in the 4-week baseline period) (Fig. 3a). This equates to 516 kg of waste per year which is no longer being autoclaved and then incinerated.
The processes adopted by the team required roughly 30 minutes three times per week to empty the decontamination station. It also required the cooperation and support of the Central Services Unit for dishwasher and autoclave use.
Find out more about how to prevent waste in your lab

Cleaning methods were tested on ‘single-use’ items
The researchers looked into two options for sterilising items that had previously been considered ‘single-use’.
- Chemical decontamination in a ‘decontamination station’ (soaking items in a 10% Distel disinfectant solution overnight – 16h+)
- Washing and autoclaving (using high pressure and heat)
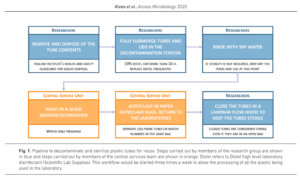
They found that one or both of these methods would produce items that were suitably sterile for a lot of their work. Work that required ‘non-pyrogenic’ conditions (I.e. Absolutely free from bacterial contamination) is still undertaken using non-pyrogenic single-use lab plastics. Similarly, alternatives have not yet been found for non-cytotoxic conditions. This limited the use of the reused items to microbiological procedures and excluded the items from tissue culture procedures. On a larger scale (I.e. building-wide) plastics could be ‘cascaded’ from areas where high levels of sterility were required (e.g. tissue culture) to areas where there is greater tolerance.
Some single-use lab plastic items were found to be robust enough for autoclaving (Falcon tubes), while others melted (universal tubes, petri dishes). For those items which melted, some could be replaced with reusable items made of other materials, such as glass or metal.
Different cleaning methods worked on different plastic lab items
The below describes the various ways the group reduced their production of plastic waste.
Plastic weighing boats
Washed, dried and reused.
Plastic serological pipettes/stripettes
When used for pipetting common non-sterile solutions (e.g. ethanol, concentrated buffered solutions) they would be sheathed in their plastic wrap, labelled with the working solution that they were used for, and placed in an agreed location for reuse.
Plastic cuvettes
Decontaminated overnight in 10% Distel, rinsed with water, dried and reused.
Plastic tip collection containers
- Contaminated tips transferred to biohazard bins when the container is full.
- Container decontaminated overnight in 10% Distel, rinsed with water, dried and reused.
Plastic Petri dishes
Petri dishes used with media only are decontaminated overnight in 10% Distel, washed and reused for agar media with antibiotics
Plastic 15 and 50ml Falcon tubes
- Chemically decontaminated overnight in 10% Distel
- Rinsed with water, washed in a dishwasher with a water-only programme,
- Autoclaved in bags.
- Tubes are closed in a cell culture hood and considered sterile for non-cell culture work.
- Stopped using universal tubes since they cannot be autoclaved for reuse.
Of note, we did not observe contamination related to the reuse of chemically decontaminated and autoclaved plastic tubes following the implementation of reuse guidelines, making us confident that the pipeline is appropriate for our research applications.
Plastic inoculation loops
- Replaced with metal inoculation loops.
- Washed and autoclaved for reuse.
Note: the inoculation loops require to be sterilised with a flame between samples, which adds a little time to procedures.
Plastic tips for bacterial colony picking
- Replaced with wooden sticks.
- Washed and autoclaved for reuse.
More opportunities
In addition, the group looked at the activities they were undertaking and considered opportunities for improvements.
Plan experiments to reduce single-use items
- Prepare master mix to reduce tips.
- Organize experimental layouts to use the minimum number of tubes and plates.
- Use 96-well plates instead of Eppendorfs for serial dilutions in experiments with several dilutions/ conditions.
- Divide the agar plate into parts (more than one condition per plate).
- Use lines and drop plating technique for experiments with bacterial serial dilutions.
- Use the same tips for the same condition.
Non-pyrogenic vs autoclaved lab plastics
- When possible, use in-house autoclaved tips (with reused tip boxes and without plastic wrapping) instead of filtered tips.
- For work that does not need to be non-pyrogenic or RNA/DNA-free, use autoclaved tubes (sterile and reused).
In general: before any protocol step ask: “is there a more sustainable way to do this?”.
The group are continuing to look for ways to improve, including investigating the introduction of glass culture tubes, Petri dishes and stripettes to replace plastic versions. These glass items can be decontaminated by autoclaving and reused without the need for a chemical decontamination station.





This is a great study which deserves wide promulgation/adoption throughout CMVM. Assume that’s happening?