Author: Holly Ennis

The National Cancer Research Institute Annual Meeting in Glasgow 2019 marked an important milestone for members of the ECTU health economics group involved in the Lothian Cancer Data Intelligence Project (LCDI). It marked the end of an 18 month collaborative project to explore the completeness, data quality and scope of a comprehensive linked cancer regional dataset accessed by NHS Lothian Analytical Service. The project, partially funded by the Association of the British Pharmaceutical Industry, Pfizer UK, Novartis Pharmaceuticals UK Ltd, AstraZeneca UK and NHS Lothian and funded through Joint Working with Roche Products Ltd, also sought to explore the need for real world data and how to facilitate data requests from different stakeholder groups.
In a final meeting the key deliverables of the study were presented: a data dictionary and data quality report in the form of a wiki within the public domain and a research governance framework managed by NHS Lothian ready to pilot data requests. An important part of the project was a series of worked examples, modelled around a relevant research question, which was used to test out the processes and instructions developed as part of the LCDI study. The largest of these research questions was pathway visualisation (featured below) using national datasets to look at treatment sequencing patterns in secondary breast cancer (ER +ve HER2 –ve). This was developed into an abstract and presented at the NCRI 2019 meeting by Dr Ashley Horne as part of the Silent Theatre!
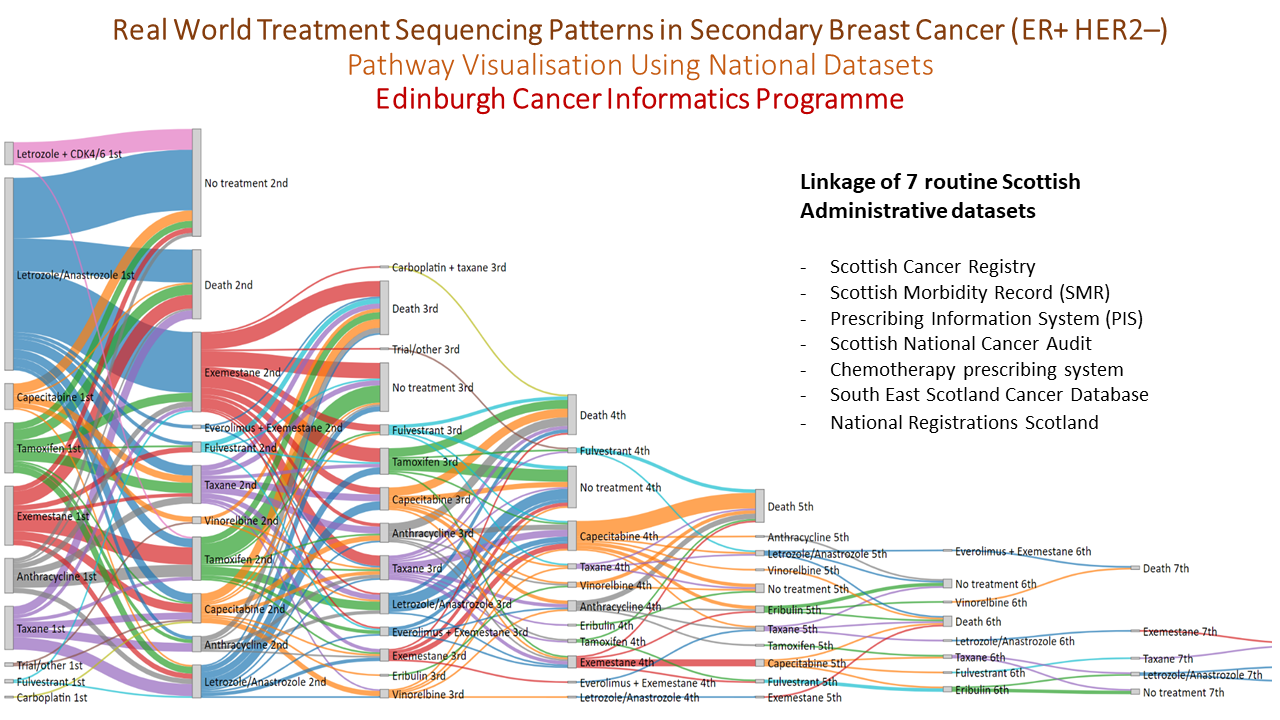
Learning from the LCDI project has led to the launch of the ‘Edinburgh Real World Data Service’ which is run jointly by the Lothian Research Safe Haven Team and the Edinburgh Cancer Informatics Programme with the objective of improving the availability and use of real NHS data to inform new reimbursement models that can ultimately provide better value medicines for NHS patients.
Learn more about the Edinburgh Real World Data Service here.



