How my brother’s diagnostic odyssey led me to a PhD
By Cléo Pereira De Almeida, PhD student
I remember swinging my legs in the waiting room chair. My brother and parents were behind a closed door that the nurses kept insisting I could not enter. Still, I could hear my mother crying. Appointment after appointment and test after test, a quiet realisation began to form: my brother had something no one could clearly explain. He underwent hundreds of examinations, received dozens of conflicting diagnoses, and tried countless medications before eventually stepping away from treatment altogether. As I grew older, I came to understand that his story was not unique. It reflected the experiences of millions of families confronting the uncertainty inherent to rare diseases.
Journeys like his are often described as a diagnostic odyssey: a prolonged and uncertain search for answers that unfolds when clinical information alone is insufficient to reach a diagnosis. Patients and their families may move through years of specialist consultations, imaging studies, and laboratory investigations, often without clear resolution (figure 1) 1,2. While this experience is deeply personal for each family, it also reflects a broader and often overlooked public health reality. Rare diseases, which encompass an estimated 5,000 to 8,000 distinct conditions, collectively affect around 475 million individuals worldwide 3. Since around 80% of rare diseases have a genetic origin, genomic insights are central to diagnosis and management 4. In Latin America, where I am from, around 40 to 50 million individuals live with a rare disease, but many face limited access to genetic tests and treatment 2. It was within this landscape of unmet needs that my desire to pursue genetics took root.
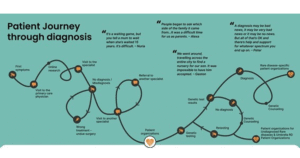
Figure 1. Diagram illustrating the patient journey to diagnosis in the context of rare diseases, as developed by the Community Engagement Task Force (CETF) of EURORDIS‑Rare Diseases Europe within the Solve‑RD project. This infographic visualizes the typical diagnostic odyssey experienced by patients, from the onset of first symptoms and early clinical encounters, through multiple referrals, misdiagnoses, and genetic testing, to eventual diagnosis, genetic counselling, and engagement with patient organisations. It highlights the complexity, delays, and emotional challenges often encountered on the path to diagnosis, alongside resources and support mechanisms available at various stages. Source: Solve‑RD infographic on the patient journey to diagnosis, EURORDIS‑Rare Diseases Europe 6.
This search for answers brought me to the Institute of Genetics and Cancer (IGC). I chose the IGC because it stands at the forefront of uniting fundamental research with clinical application. This is exactly what is needed to solve diagnostic puzzles like the one my brother faced. As part of the MRC Human Genetics Unit PhD programme, the first year involves laboratory rotations that allow us to explore different research avenues. For my first rotation, mentored by Dr Andrew Jackson and Dr Martin Reijns, I focused on somatic mutagenesis. This project resonated with me because it directly relates to mosaicism, defined as the presence of two or more cell lineages with different genotypes arising from a single zygote 5. I believe mosaicism remains an underestimated factor in the global burden of genetic diseases, and since I have always been drawn to scientific questions that are often overlooked, it is a topic I hope to address in my final PhD project. Currently, I am undertaking my second rotation with Dr Martin Taylor, where we are applying machine learning models to identify DNA alterations using nanopore sequencing. This journey through rotations has only reinforced my conviction that advancing our understanding of complex genetic phenomena is essential not just for scientific discovery but for driving real improvements in diagnosis, care and equity in global health outcomes.
Ultimately, my goal is to ensure that the closed doors my family faced become open pathways for others. While the past cannot be rewritten, my work at the IGC empowers me to shape a more certain future for families seeking answers. By investigating the underexplored mechanisms of the genome, I aim to transform unexplained symptoms into clear diagnoses, reducing the burden of uncertainty for the next generation of patients.
References
- Wu AC, McMahon P, Lu C. Ending the Diagnostic Odyssey-Is Whole-Genome Sequencing the Answer?. JAMA Pediatr. 2020;174(9):821-822. doi:10.1001/jamapediatrics.2020.1522
- Velasco HM, Bertoli-Avella A, Jaramillo CJ, et al. Facing the challenges to shorten the diagnostic odyssey: first Whole Genome Sequencing experience of a Colombian cohort with suspected rare diseases. Eur J Hum Genet. 2024;32(10):1327-1337. doi:10.1038/s41431-024-01609-8
- Abozaid GM, Kerr K, Alomary H, Al-Omar HA, McKnight A. Global insight into rare disease and orphan drug definitions: a systematic literature review. BMJ Open. 2025;15(1):e086527. Published 2025 Jan 25. doi:10.1136/bmjopen-2024-086527
- Gürkan H, Bilge Satkın N. The Importance of Genetic Diagnosis in Rare Diseases. Balkan Med J. 2025;42(2):92-93. doi:10.4274/balkanmedj.galenos.2025.2025-270125
- Geiger H, Furuta Y, van Wyk S, Phillips JA 3rd, Tinker RJ. The Clinical Spectrum of Mosaic Genetic Disease. Genes (Basel). 2024;15(10):1240. Published 2024 Sep 24. doi:10.3390/genes15101240
- EURORDIS – Solve-RD Infographic on the Patient Journey to Diagnosis. Available at: https://www.eurordis.org/publications/solve-rd-infographic-on-the-patient-journey-to-diagnosis.



