The impact of spatial omics: a transformative technology in biomedical research
By Yuqin Wang, PhD student
Life is built upon cells, the basic units of all living organisms, much like bricks form the foundation of buildings. To gain deeper insights into cellular structure, function and interactions, a wide range of technologies is used in biomedical research to unravel the complex mechanisms underlying living organisms.
These methodologies include polymerase chain reaction (PCR), DNA/RNA sequencing, CRISPR-Cas9 gene editing, cell culture, western blotting, immunohistochemistry, and many others. For decades, researchers have relied on bulk analyses that average signals from thousands to millions of cells to generate valuable datasets.
While these approaches have enabled us to understand the complex biology, they inevitably overlook cellular heterogeneity. As technology has evolved, spatial omics has emerged as a powerful innovation that bridges this gap by preserving the spatial architecture of tissues while providing high-resolution molecular information.
What Is Spatial Omics?
Spatial omics is an umbrella term which includes a range of technologies, including spatial transcriptomics, spatial proteomics, spatial epigenomics and spatial metabolomics. These innovative approaches allow researchers not only to characterise molecular profiling and interactions across cells but also to map their precise locations within intact tissues simultaneously.
By integrating spatial approaches with other advanced technologies, researchers can achieve a more comprehensive and accurate understanding of biological systems. For example, single-cell RNA sequencing (scRNA-seq) allows researchers to measure gene expression at the individual cell level; however, it requires the disruption of intact tissue sample, resulting in the loss of spatial context, cellular interactions and the tissue structure.
In contrast, spatial transcriptomics is capable of analysing gene expression patterns in situ while preserving tissue architecture. When combined with next-generation sequencing (NGS) or imaging-based approaches (e.g., fluorescence in situ hybridisation (FISH) and in situ sequencing), it enables visualisation and quantification of transcripts at high spatial resolution, sometimes even at single-molecule levels [1].
At the protein level, spatial proteomics based on mass spectrometry (MS) or advanced imaging techniques (e.g., imaging mass cytometry (IMC), multiplexed ion beam imaging (MIBI)), enables us to map the protein distributions, functions and interactions across the tissues [2, 3]. This provides critical functional insights, as proteins are the primary executors of cellular processes.
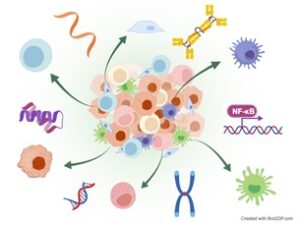
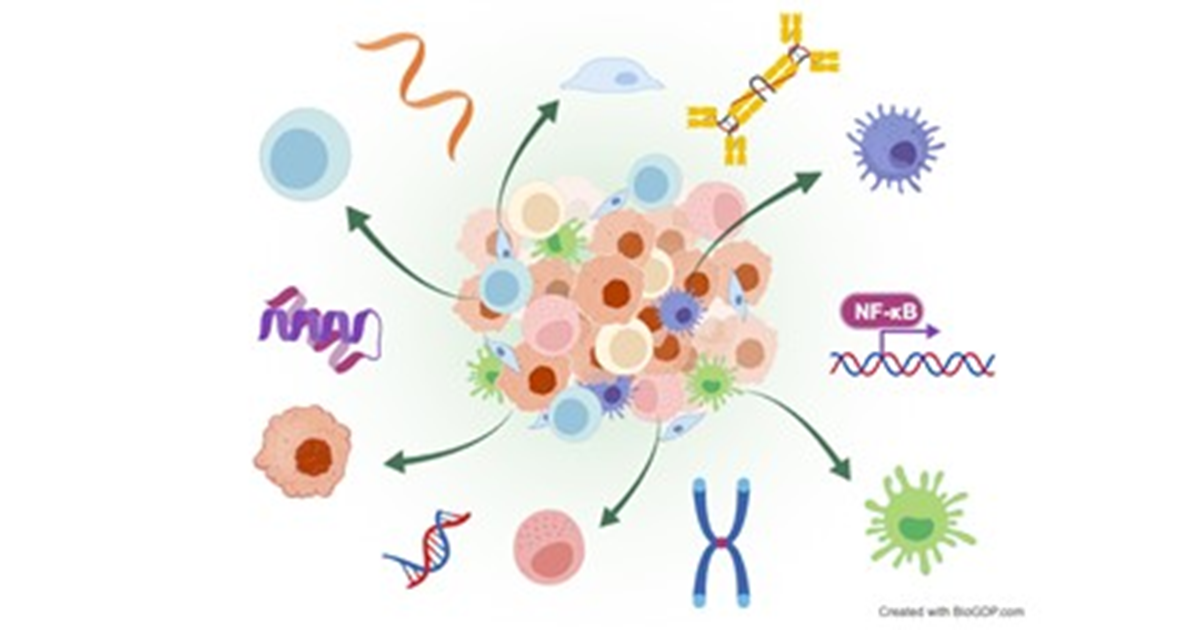
Application in Clinical Research
Spatial omics technology has demonstrated particular promise in cancer research. Solid tumours, such as colorectal cancer, breast cancer, prostate cancer and melanoma, are not merely masses of tumour cells; instead, they also consist of diverse cell populations, including immune cells, stromal cells and vasculature, which collectively form a complex ecosystem known as the tumour microenvironment (TME).
Recently, multiple studies have applied spatial transcriptomics in combination with scRNA-seq in colorectal cancer. Researchers have deciphered the spatially-resolved cellular and molecular composition of colorectal cancer and mapped the intratumoral heterogeneity of Consensus Molecular Subtypes and their associated microenvironments, highlighting how spatial transcriptomics can refine our understanding of tumour heterogeneity and support the development of personalised therapies [4].
Beyond oncology, spatial omics is increasingly being applied to non-neoplastic diseases, such as neurological diseases and autoimmune diseases. In Alzheimer’s disease, spatial omics helps investigate molecular alterations directly within intact brain tissue, which clarifies cellular interactions and signaling pathways associated with disease progression and pathogenesis, while simultaneously minimising sample loss [5].
In autoimmune diseases such as systemic sclerosis, lupus nephritis, rheumatoid arthritis and autoimmune thyroid disease, spatial omics has dissected disease-specific immune landscapes and molecular profilings, offering new microscopic perspectives on disease mechanisms.
Future Directions
New technologies continue to reshape the landscape of biomedical research, and spatial omics stands out as a transformative breakthrough. Nevertheless, the field is still in its early era. Several challenges remain, including high equipment costs, the need for standardised analytical pipelines and the difficulty of interpreting large-scale, high-dimensional datasets.
Integrating spatial omics with deep learning frameworks may provide effective solutions to these challenges. As more laboratories gain access to these new-generation techniques, spatial omics is ready to unlock the incredible biological insights.
References:
[1] Lee, Y, Lee, M, Shin, Y, et al., Spatial Omics in Clinical Research: A Comprehensive Review of Technologies and Guidelines for Applications. Int. J. Mol. Sci. 2025, 26, 3949.
[2] Wu M, Tao H, Xu T, et al., Spatial proteomics: unveiling the multidimensional landscape of protein localization in human diseases. Proteome Sci. 2024, 20;22:7.
[3] Xu Y, Lih TM, De Marzo AM, et al., SPOT: spatial proteomics through on-site tissue-protein-labeling. Clinical Proteomics, 2024, 21, 60.
[4] Valdeolivas A, Amberg B, Giroud N, et al., Profiling the heterogeneity of colorectal cancer consensus molecular subtypes using spatial transcriptomics. npj Precis. Onc. 2024, 8, 10.
[5] Marshall CR, Farrow MA, Djambazova KV, et al., Untangling Alzheimer’s disease with spatial multi-omics: a brief review. Front. Aging Neurosci. 2023, 15:1150512.



